※ This post is content for providing medical information.
It is not subject to review under Article 56, Paragraph 2 and Article 57 of the Medical Service Act,
and is not a post for advertising purposes.
Oral Wegovy? Wegovy Pills?
Could it be used in Korea?
Hello,
I am Sung Hyun-joo, director of Cheongdam OK Clinic.

| ▶ Regular member of the Korean Academy of Cosmetic Surgery (KACS) |
|---|
| ▶ Regular member of the Korean Society for Obesity (KSSO) |
| ▶ Regular member of the Korean Association for the Study of Obesity and Aesthetics (KAAM) |
| ▶ Regular member of the Korean Society for Liposuction Surgery (KSSL) |
| ▶ Regular member of the Korean Society of Aesthetic Surgery (KSAS) |
Recently, the expressions “oral Wegovy” and “Wegovy pills”
have been spreading rapidly online.
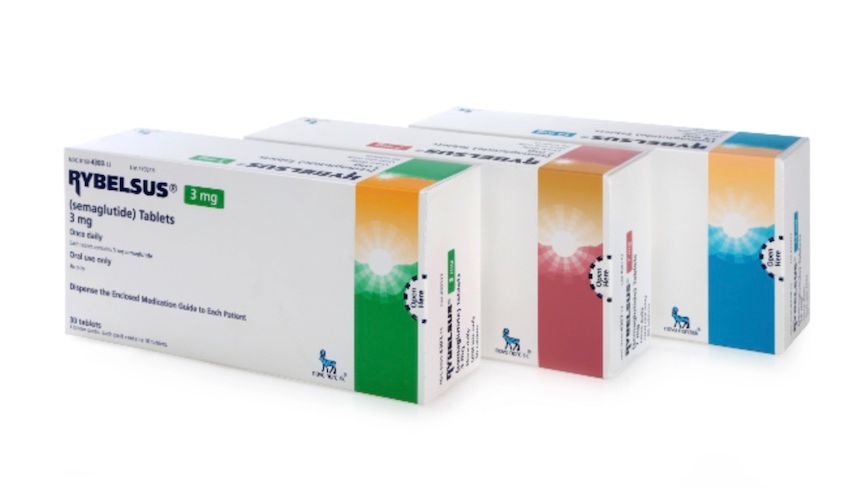
However, in Korea, an oral form of Wegovy
is not currently approved or distributed.
Some sales pages promote it as a pill form,
but

there have been reports of products that are not officially approved,
so caution is needed.
Products that have not gone through official distribution channels make it difficult to verify their ingredients and dosage,
and there is a risk of unexpected side effects.

Therefore, at this point, claims that “oral Wegovy” is being legally sold on the market
are not true.
Principles and absorption structure of oral GLP-1 formulations
Oral GLP-1 class drugs already exist.
 Oral Wegovy
Oral Wegovy
A representative example is Rybelsus, whose active ingredient is semaglutide,
and
it is a product approved by the U.S. Food and Drug Administration (FDA).
However, this drug was approved not for obesity treatment,
but for the management of type 2 diabetes.

Because GLP-1 is originally a peptide structure that is easily broken down in the digestive tract,
its oral absorption rate is very low.
In fact, the absorption rate is known to be less than 1%.
For this reason, a much higher oral dose is needed to achieve blood concentrations similar to those of an injectable formulation.
It is not simply a matter of “switching to a pill”;
you need to understand that the drug delivery method itself is different.
Release status and possibility of domestic introduction
A high-dose oral semaglutide formulation
was approved by the U.S. FDA in December 2025 for the obesity treatment indication,

and the launch schedule in the United States began in January 2026.
However, approval and launch in Korea have not yet been confirmed.
Introducing a drug requires several procedures, including review by the Ministry of Food and Drug Safety, reimbursement listing, and distribution contracts.

Typically, it can take from several months to more than a year from overseas approval to domestic approval.
Therefore, information claiming that “oral Wegovy” is officially being sold in Korea at present
is not true.
Whether it will be introduced in the future will depend on the pharmaceutical company’s application and the regulatory authority’s review schedule.
Price outlook and comparison of efficacy and side effects
Oral semaglutide is a prescription drug,
and there is currently no formulation approved in Korea for the obesity indication.

The timing of introduction and the price are undecided, and the actual out-of-pocket cost may vary depending on whether insurance coverage is applied.
In clinical studies, meaningful weight loss has been reported with high-dose administration.

However, gastrointestinal side effects such as nausea, vomiting, and diarrhea may occur,
and the frequency may increase as the dose increases.
Convenience of taking the medication is an advantage, but due to the low absorption rate,
a higher-dose strategy is required.
-Clinic hours-
| Weekdays 10:00 ~ 19:00 (Reception closes at 18:30) Saturday 10:00 ~ 16:00 (Reception closes at 15:30) Closed on public holidays / Wednesdays and Sundays |
-Contact-
| ▶ Cheongdam OK Clinic |
|---|
| Consultation phone: 02-3446-7588 KakaoTalk inquiry |
-Directions-
Oral Wegovy




 Oral Wegovy
Oral Wegovy




