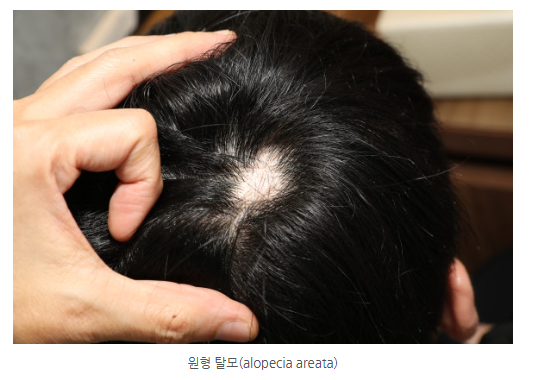
When patients hear that a new drug may be effective for severe alopecia areata, many feel hopeful on the one hand, but on the other hand, they find it hard to believe.
That is because many people have tried various treatments over the years, only to see no clear results or to experience recurrence.
So while news of a new treatment is welcome, it is also understandable to take a step back and watch cautiously.
The clinical results of the newly introduced “Linvio® (upadacitinib)” that I will introduce today are also a case to consider with that kind of cautious optimism.
AbbVie, the global pharmaceutical company, has announced meaningful Phase 3 clinical results for Linvio® (active ingredient: upadacitinib) in severe alopecia areata.
Linvio® was originally used for atopic dermatitis and rheumatoid arthritis,
and in this study of hair loss patients as well, it confirmed
hair regrowth.
The scalp of the patients enrolled in the trial had, on average, only about 16% hair remaining.
However, after 24 weeks, more than half of the patients who took Linvio® 30 mg experienced hair regrowth on more than 80% of the scalp.
Even more remarkably, nearly half of the patients recovered to more than 90%.
JAK inhibitors such as baricitinib and ritlecitinib have already drawn attention by making their names known in the alopecia areata treatment market, but the results with Linvio® show an even stronger and faster response.
In particular, the fact that improvement in the eyebrows and eyelashes was also observed can be especially meaningful for patients who hope for more than just the “aesthetic recovery” of hair loss.
Of course, concerns about side effects come along with the drug’s benefits.
Some serious infections were reported in the group that received Linvio®.
This is related to the drug’s mechanism of action.
Upadacitinib is an immunomodulator that selectively inhibits JAK1.
JAK1 is involved in several cytokine signaling pathways that mediate inflammatory responses in the body, and inhibiting this pathway has the advantage of protecting hair follicles from autoimmune reactions.
However, because it also partially blocks the immune response itself, immune defense against bacteria and viruses may be reduced.
In other words, hair may begin to regrow, but in some patients immune suppression may occur and, rarely, the risk of infection may increase.
Fortunately, no fatal infections or serious complications were observed in this clinical trial, and most adverse events were manageable, such as acne, nasopharyngitis, and mild upper respiratory infections.
Professor Arash Mostaghimi of Brigham and Women’s Hospital said that “alopecia areata is often not simply a cosmetic issue, but a condition that can affect mental health,” and stated that this Linvio® study shows the possibility of a true treatment.
In fact, many alopecia areata patients I meet in the clinic cannot easily believe it even after hearing that “hair grows back.”
Hair loss can be triggered by genetic factors or stress, but recovery is as uncertain as the direction of the wind, so people become cautious even about having hope.
But this study is at least gradually reducing some of that uncertainty.
Of course, it is still unknown when Linvio will be officially approved in Korea as a treatment for alopecia areata.
However, at least for now, we can speak more confidently about the word “possibility.”
And I hope these research results can also be meaningfully used in actual clinical settings.
Of course, it has not yet been approved in Korea, and more long-term safety data are needed.
Still, for patients with severe alopecia areata who have limited treatment options, the fact that it could become a new option is itself an important step forward.
Hair loss is often dismissed as only an appearance issue, but for patients it is a condition that greatly affects quality of life.
I hope related studies will continue so that more treatment options can be developed and applied in real-world care.

Now it is time for hair growth, this has been Kim Jin-oh.
May new hair be born. (Bi-saeng-sin-mo, 必生新毛.)
References
-
He, X. et al. (2024) ‘Upadacitinib for alopecia areata in different backgrounds: a case series’, Clinical, Cosmetic and Investigational Dermatology, 17, pp. 123–130. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10936532 (Accessed: 2 August 2025).
-
Flora, A., Kozera, E. and Frew, J.W. (2023) ‘Treatment of alopecia areata with the Janus kinase inhibitor upadacitinib: a retrospective cohort study’, Journal of the American Academy of Dermatology, 89(1), pp. 137–138. DOI: 10.1016/j.jaad.2022.12.056 (Accessed: 2 August 2025). Available at: https://www.jaad.org/article/S0190-9622%2823%2900191-3/fulltext
-
Picone, V. et al. (2025) ‘Upadacitinib for the management of severe alopecia areata in adolescent patients: a single centre retrospective study’, Clinical and Experimental Dermatology, 50(1), pp. 153–160. DOI: 10.1093/ced/zkad025 (Accessed: 2 August 2025). Available at: https://academic.oup.com/ced/article-abstract/50/1/153/7726807
[This post is being written directly by a plastic surgery specialist for informational purposes in accordance with Article 56, Paragraph 1 of the Medical Service Act. Hair transplant surgery and treatment may have side effects, and please make a careful decision after consulting with a specialist.]


