Oral dutasteride is already widely used in the treatment of male pattern hair loss.
However, to reduce hormone changes and the possibility of side effects caused by systemic absorption,
development of a topical form that delivers the drug directly to the scalp is underway.
Topical preparations have the advantage of concentrating the drug effect in the target area while minimizing systemic exposure, and several clinical trials have been conducted in recent years.

A recently published phase II clinical trial compared the efficacy of topical dutasteride 0.05% with oral finasteride 1 mg.
A phase II clinical trial is the stage in which a drug’s efficacy and safety are formally evaluated,
and it is still before market launch.
For reference, phase I confirms safety, phase II evaluates efficacy and the appropriate dose, and phase III verifies efficacy and side effects on a larger scale before approval is obtained.
This study involved 135 male patients with androgenetic alopecia aged 20–60 years
and compared topical dutasteride (0.01%, 0.02%, 0.05%), oral finasteride 1 mg, and placebo.
The topical preparation was applied once daily at 1 mL to an approximately 1.9 cm² area of the crown and kept in place for 6–8 hours.
The results were compared using crown hair count, hair thickness, investigator photographic assessment, and patient questionnaires.
Looking at the results, over 24 weeks the number of hairs within 1 cm² of the crown increased by +75.52 with the 0.05% topical preparation, +41.21 with finasteride, and +0.07 with placebo.
The difference between the 0.05% topical preparation and finasteride was +34.30, which was statistically significant (p=0.0083).
Hair thickness improved in both the 0.05% topical preparation group (+11.59) and the finasteride group (+10.68) compared with placebo, but there was no significant difference between the two groups.
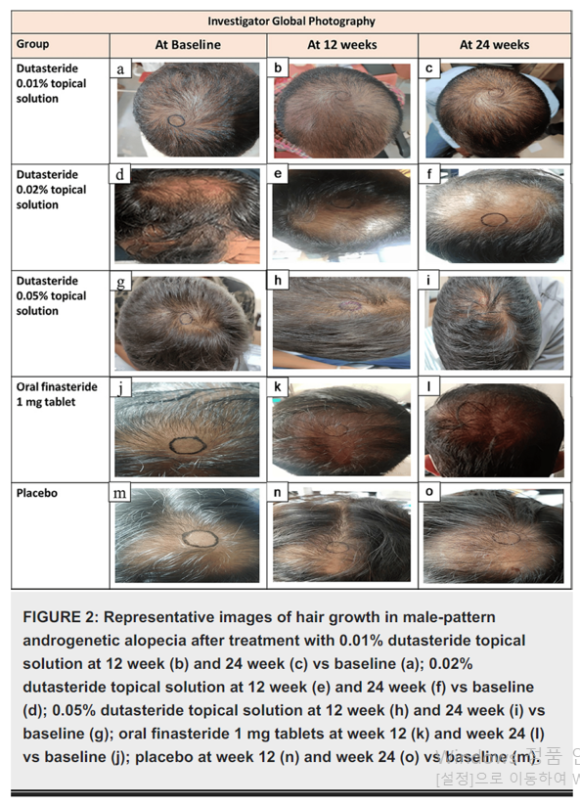
Photo source Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Alluri, C.V., Mohammad, J., KSSVV, S.R. and Dundigalla, M.R. (2025) ‘A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects With Androgenetic Alopecia’, Cureus, 17(8), e89309
In the photographic assessment, the proportion rated as “moderate or greater increase” (≥+2) was 68.97% for the 0.05% topical preparation, 21.43% for finasteride, and 13.33% for placebo.
In the patient questionnaire, satisfaction with the crown-related item (score ≤2) was 96.55% for the 0.05% topical preparation and 71.43% for finasteride. Both patient perception and photographic assessment favored the topical preparation.
In terms of safety, there were no serious adverse events or treatment discontinuations,
and most skin irritation was rated as “none.”
Hormonal changes were also smaller with the topical preparation than with the oral agent.
In pharmacokinetic analysis, the 0.05% topical preparation reached the highest blood concentration about 5 hours after application, but overall levels were very low.
This study has limitations, including evaluation focused on the target area, a 24-week short-term observation period, and a population composed mainly of Indian men. The product has not yet been launched, and phase III clinical trials and long-term observation are needed for commercialization.
If phase III is completed in the future and safety is confirmed, there is a high possibility that it will be launched.
I will wait for that time.
Item Summary
| Item | Summary |
|---|
| Study purpose | Compare the efficacy of topical dutasteride (0.01%, 0.02%, 0.05%) and oral finasteride |
| Study phase | Phase II clinical trial (not yet on the market; phase III needed) |
| Participants | 135 male AGA patients aged 20–60 years |
| Dosing method | Topical: 1 mL daily applied to a 1.9 cm² crown area and kept for 6–8 hours / Oral: finasteride 1 mg once daily |
| Main endpoints | Hair count (TAHC), hair thickness (TAHW), investigator photographic assessment (GPA), patient satisfaction (MHGQ) |
| Main results | 0.05% topical TAHC +75.52, finasteride +41.21, placebo +0.07 / GPA ≥+2: 0.05% 68.97%, finasteride 21.43% / MHGQ ≤2: 0.05% 96.55%, finasteride 71.43% |
| By concentration | 0.05% showed the greatest effect; 0.02% and 0.01% also improved versus placebo / Superiority over oral treatment reached statistical significance only for 0.05% |
| Comparison | Statistical significance was confirmed only for 0.05% |
| Safety | No serious adverse events, little skin irritation, mild hormonal changes |
| Conclusion | The 0.05% topical preparation was most favorable in terms of efficacy and safety; long-term safety needs to be confirmed in phase III clinical trials |

It is now time for hairhair, Kim Jin-oh.
May new hair be born.
References
Panuganti, V.K., Kumar Madala, P., Ramalingayya Grandhi, V., Alluri, C.V., Mohammad, J., KSSVV, S.R. and Dundigalla, M.R. (2025) ‘A Randomized, Double-Blind, Placebo and Active Controlled Phase II Study to Evaluate the Safety and Efficacy of Novel Dutasteride Topical Solution (0.01%, 0.02%, and 0.05% w/v) in Male Subjects With Androgenetic Alopecia’, Cureus, 17(8), e89309.
[This post is written directly by a board-certified plastic surgeon for informational purposes in accordance with Article 56, Paragraph 1 of the Medical Service Act. Hair-loss surgery and treatment may involve side effects, so please make a careful decision after consulting with a specialist.]


