Baricitinib
Source - Medical Times
Ritlecitinib
Source - Good Morning Chungcheong
Upadacitinib
Source - Real News
Severe alopecia areata is an immune disorder that affects more than just hair loss; it has a major impact on appearance, self-esteem, and social life.
In the past, treatment was limited to saying, let’s just watch and wait for natural recovery.
But in recent years, the treatment environment has fundamentally changed.
With the emergence of JAK inhibitor–based targeted therapies such as baricitinib, ritlecitinib, and upadacitinib, severe alopecia areata is now being recognized as a condition for which a treatment strategy can be designed.
Alopecia areata: Has treatment changed since baricitinib, ritlecitinib, and upadacitinib?
Summary
Severe alopecia areata is an autoimmune disease caused by breakdown of immune privilege
Targeted therapies that block the JAK-STAT pathway have emerged
With baricitinib, ritlecitinib, and upadacitinib, meaningful recovery of 40–50%
or more is possible
Because relapse can occur after stopping treatment, maintenance therapy strategies are important
Currently not covered by insurance in Korea, but discussions about insurance coverage are underway
Q1. Even if alopecia areata is severe, can hair really grow back?
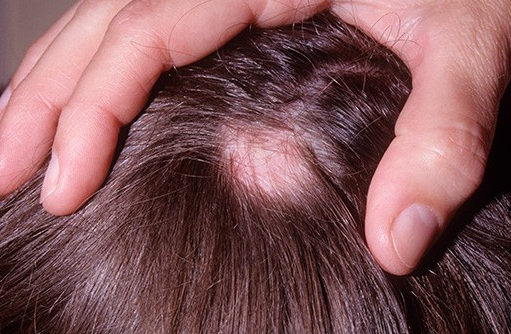
Alopecia areata
Source - Hidoc
Unlike in the past, we are now at a stage where this possibility can be discussed.
In large clinical trials involving patients with severe alopecia areata, use of JAK inhibitors has repeatedly shown that about 35–50% of patients experienced substantial recovery of the scalp¹⁴⁸.
In clinical practice as well, some patients with near-total scalp hair loss or near-universal hair loss experience a clear recovery process within a few months, progressing from fine vellus hair to thicker terminal hair.
We cannot guarantee the same outcome for every patient, but this is clearly different from an era when nothing could be done.
Q2. Why do some people improve quickly while others respond less well?
The course of alopecia areata varies greatly depending on when and how strongly immune privilege breaks down, as well as a person’s genetic background¹.
Clinically, shorter disease duration, younger age, and a history of response to previous treatment tend to be associated with a better treatment response⁷.
This is interpreted to mean that recovery is more likely when the pathway is blocked before the immune attack becomes entrenched.
Q3. How do JAK inhibitors work?
In alopecia areata, inflammatory signals such as interferon-gamma and interleukin-15 are amplified through the JAK-STAT pathway¹².
Baricitinib, ritlecitinib, and upadacitinib selectively block this pathway and help stop excessive immune attacks around the hair follicles²⁴.
They are classified as targeted therapies because they do more than simply suppress inflammation; they help rebalance the disrupted immune environment.
Q4. When does treatment effect appear?
The timing varies depending on the medication and the individual’s condition,
-
In rapid responders, an increase in vellus hair may appear within 1–3 months
-
Visible changes in density often appear by months 4–6
-
Stabilization often occurs by months 9–12
This pattern is commonly seen⁸.
The important point is not to stop too hastily even if the initial response is slow.
Long-term follow-up studies have shown a tendency for recovery rates to increase the longer treatment is maintained⁵.
Q5. If you stop the medication, will the hair fall out again?
Yes, relapse was observed in many patients within 6 months after treatment was discontinued⁶.
That is why recent treatment strategies recommend a stepwise approach such as allowing sufficient recovery, switching to a maintenance dose, and attempting dose reduction while monitoring relapse risk.
This can be understood as a concept similar to treatment for hypertension or diabetes.
Q6. How much is the cost burden?
Currently in Korea, all of these are non-covered items.
Medication cost: around 600,000 KRW per month
With tests included: often felt to be around 700,000–800,000 KRW per month.
However, discussions about health insurance coverage have recently become active, and the possibility of reimbursement for severe patients is also being discussed in practical terms.
Alopecia areata: Has treatment changed since baricitinib, ritlecitinib, and upadacitinib? Summary table
| Drug name | Approved age | Main response rate | Features |
|---|
| Baricitinib | Adults | About 35–40% | First approved, abundant long-term data³⁴ |
| Ritlecitinib | Age 12 and older | About 40% | Can be used in adolescents⁷ |
| Upadacitinib | Adults | Initial response over 50% | Rapid effects reported⁸⁹ |
Severe alopecia areata is no longer a disease left to chance.
It is changing into a condition that can be planned for—setting treatment goals, choosing medications, and considering maintenance strategies.
This change is not simply the arrival of a new drug, but rather a sign that medicine’s perspective on alopecia areata has advanced another step.
Now it’s time for Hair, Kim Jino.
Yeongsaengsinmo (必生新毛).

Written by: Kim Jino of New Hair Plastic Surgery Clinic (Public Relations Director, Korean Association of Plastic Surgeons / Academic Director, Korean Society of Laser Dermatology and Hair)
References
-
King, B., Ohyama, M., Kwon, O., et al. (2022). Two Phase 3 Trials of Baricitinib for Alopecia Areata. New England Journal of Medicine, 386(18), 1687–1699.
-
Senna, M. M., King, B., Kwon, O., et al. (2024). Long-term efficacy and safety of baricitinib in patients with severe alopecia areata. Journal of the European Academy of Dermatology and Venereology, 38(5), e603–e613.
-
US Food and Drug Administration. (2022). FDA approves baricitinib for severe alopecia areata in adults.
-
Sanchez, K., & Senna, M. M. (2025). Evaluating current and emergent JAK inhibitors for alopecia areata. Dermatology and Therapy, 15, 1–18.
-
Senna, M. M., et al. (2024). Long-term extension study of baricitinib in severe alopecia areata. Journal of the European Academy of Dermatology and Venereology.
-
King, B., Kwon, O., Guttman-Yassky, E., et al. (2024). Baricitinib Withdrawal and Retreatment in Patients With Severe Alopecia Areata. JAMA Dermatology, 160(5), 531–540.
-
King, B., Guttman-Yassky, E., Peeva, E., et al. (2023). Efficacy and safety of ritlecitinib in adults and adolescents: a phase 2b–3 trial. The Lancet, 401(10374), 1519–1531.
-
Battilotti, C., et al. (2025). Upadacitinib for alopecia: current evidence and clinical perspectives. Skin Appendage Disorders, 11(4), 320–332.
-
AbbVie Inc. (2025). Topline results from the UP-AA trial evaluating upadacitinib.
-
Javed, T., et al. (2024). FDA approval of deuruxolitinib: a new treatment option for severe alopecia areata. Journal of Clinical and Aesthetic Dermatology, 17(9), e123–e129.
-
National Alopecia Areata Foundation. (2025). FDA-approved oral JAK inhibitors for severe alopecia areata.
[This post was written directly by a board-certified plastic surgeon for the purpose of providing information, in accordance with Article 56(1) of the Medical Service Act. Hair-loss surgery and treatment may have side effects, and they should be carefully decided upon through consultation with a specialist.]




